Drug Pricing
Our work in Drug Pricing
-
Create a U.S. Institute of Health Technology Assessment to Preserve Innovation and Control Costs
Americans need affordable, accessible and innovative health care. An important first step that can be taken without risking heavy-handed intervention is for Congress to establish an independent Institute of Health Technology Assessment.
Categorized in -
USC Schaeffer Center and Aspen Institute Advisory Panel Recommend Ways to Modernize Health Technology Assessment in the U.S.
The U.S. has no public organization dedicated to producing and evaluating HTAs despite the potential of HTAs providing critical input about price and value, while also recognizing the contribution of innovation to advancing health and healthcare.
-
Health Technology Assessment in the U.S. – A Vision for the Future
Health technology assessment (HTA) can help achieve the dual health policy goals of ensuring affordability and encouraging innovation.
Categorized in -
Monthly Cost Sharing Doubles Throughout the Year for Some Medicare Insulin Users
Schaeffer Center researchers examined how use of insulin changes as cost-sharing fluctuates over the course of the year for Medicare Part D beneficiaries.
-
Without Reform, Medicare Policies Will Hamstring New Gene Therapies
It took until the fall of 2020 – three years after FDA approval – for Medicare to consistently pay an adequate amount for CAR-T therapy.
Categorized in -
Paying for Cell and Gene Therapies in Medicare
Join Schaeffer Center Director Dana Goldman in conversation with Jeet Guram and Kathy Buto about policy lessons learned and potential solutions to support innovation and patient access in cell and gene therapies.
Categorized in -
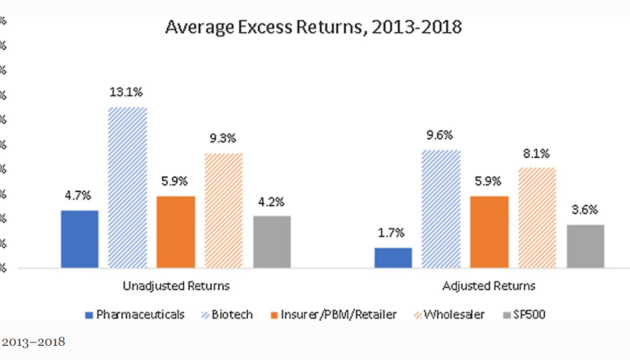
Do Companies in the Pharmaceutical Supply Chain Earn Excess Returns?
Public policies that promote competition in all areas of the pharmaceutical supply chain are important avenues for curtailing drug spending.
Categorized in -
Blood-Based Multicancer Tests Could Help Reduce Racial and Socioeconomic Cancer Health Disparities
Out of the 1.3 million individuals between the ages of 50-79 who are diagnosed with cancer, only 15% of those cases will be caught early. Aspen Institute and USC Schaeffer Center partnered for an expert discussion on early detection technologies.
-
Will the Rebate Rule need Extra Innings?
The publication of the final rebate rule is an important milestone in this administration’s drug pricing journey. However, many challenges remain.
Categorized in -
Making Progress in Cancer Diagnostics: Clinical Practice and Policy
Join the Health, Medicine and Society Program of the Aspen Institute and the USC Schaeffer Center for a webinar on new clinical advances in cancer screening and the policy issues surrounding access to and adoption of these advances.